-
March 05, 2026
-
Global New Light of Myanmar
-
55
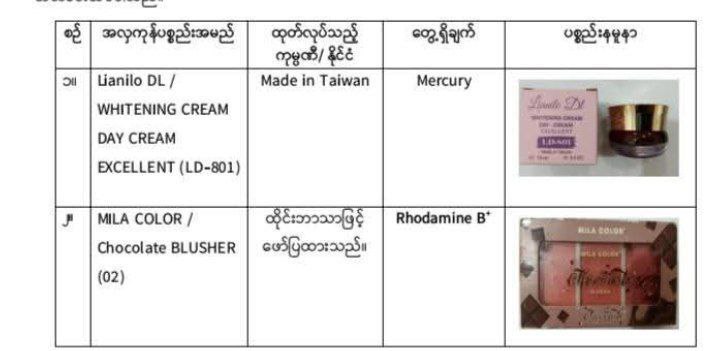
The two cosmetic products identified by the FDA as unfit for public use due to the presence of prohibited chemicals.
The Food and Drug Administration (FDA) has announced that two cosmetic products currently sold in the local market are unfit for use as they contain prohibited chemicals that pose significant health risks.
Following inspections, the FDA issued an alert on 5 March and the two products are Lianilo DL / Whitening Cream Day Cream Excellent (LD-801) made in Taiwan and MILA COLIRI / Chocolate Blusher (02) libelled in Thai. Potential side effects include skin irritation, swelling, red or white patches, numbness via skin absorption, tremors, fatigue, headaches, and depression. Chronic use can lead to kidney failure and other serious health conditions.
The FDA has explicitly warned importers, distributors, wholesalers, and retailers to adhere to professional ethics and immediately cease the sale and distribution of these unregistered and unsafe products. Failure to comply will result in legal action taken by relevant authorities under section 3, subsection (3)(d) of the Public Health Law.
Under the guidance of the Ministry of Health, the FDA continues to systematically monitor and regulate cosmetic products to ensure that the public has access to items that are authentic, high-quality, and safe for health.
MT/ZN